|
4/4/2023 0 Comments Atomic packing fraction of bcc 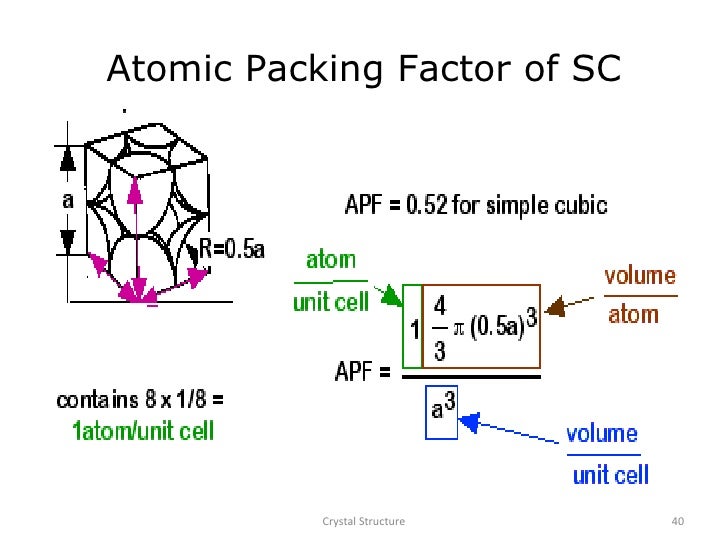
For fcc and hcp structures, the atomic packing factor is 0.74, which is the maximum packing possible for spheres all having the same diameter. 
The majority of metals take on either the hcp, ccp or bcc structure. Body Centered Cubic (BCC) Structure: Each atom has 8 equidistant nearest neighbors. The packing factors of slip systems include: Hence atoms occupy 52.35 off the volume of the unit cell. The radius of the spheres is taken to be the maximal value such that the atoms do not overlap.Ī slip plane with the highest atomic density is preferred for deformation because the distance between atoms are so small that dislocation movement due to lower applied stress is easier and higher atomic packing factor is an indication of ease of deformation. 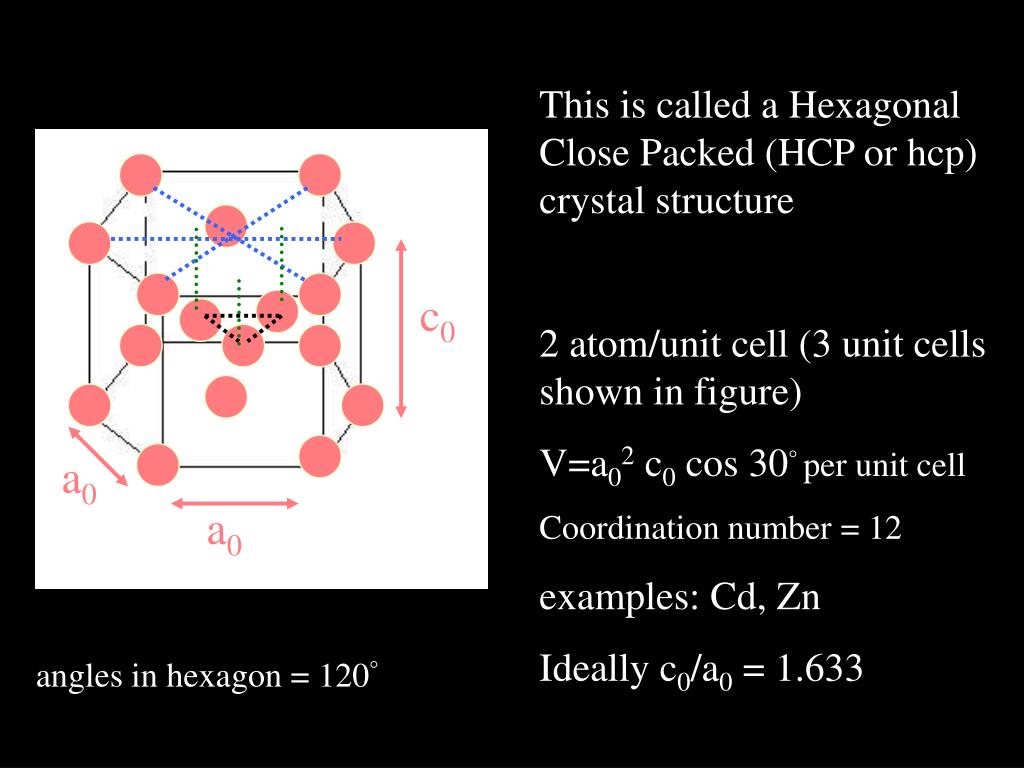
In atomic systems, by convention, the packing factor is determined by assuming that atoms are rigid spheres. Atomic packing factor (APF), packing efficiency or packing fraction is the fraction of. 
Packing factor indicates how closely atoms are packed in a unit cell and is given by the ratio of volume of atoms in the unit cell and volume of the unit cell. Atomic Packing factor for SC BCC FCC and HCP calculate or formula. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
